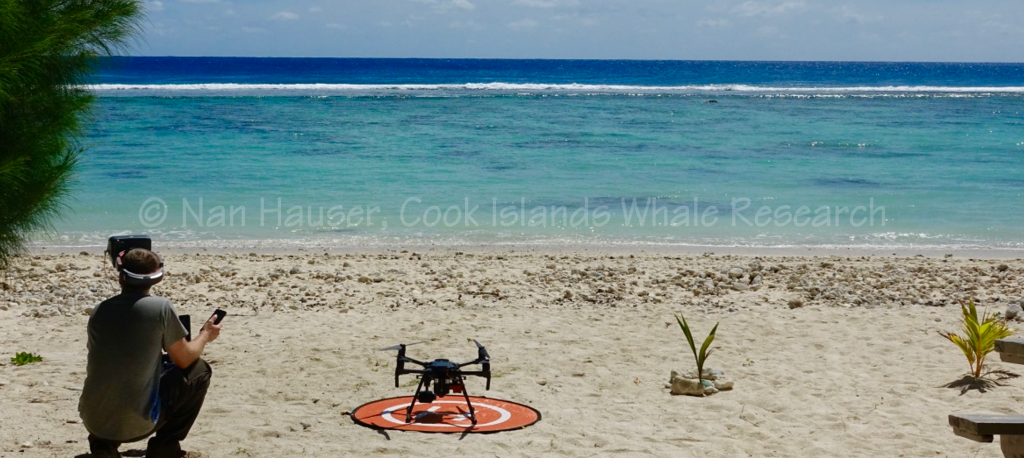Research
Since 1998, Cook Islands Whale Research (CIWR), in affiliation with Center for Cetacean Research and Conservation (CCRC), has been studying and distributing information on the populations of cetaceans and other marine mammals in the Cook Islands, South Pacific. CIWR documents population abundance of humpback whales, beaked whales, sperm whales, dolphins, other cetaceans, marine mammals, turtles and sharks through opportunistic and systematic surveys.
Every sighting is recorded with a latitude and longitude and later used for Geographic Information Systems (GIS). DNA samples of these animals are collected in order to understand population dynamics, group structures, habitat use, genotype matching and migratory pathways. The specific approaches and techniques used to perform CIWR’s research include photo-identification; acoustic analyses; satellite tagging; collection of surface, underwater and drone video footage; and collection of DNA samples for genetics, microbiology and stable isotope analysis. The research has resulted in over 65 published papers to date.
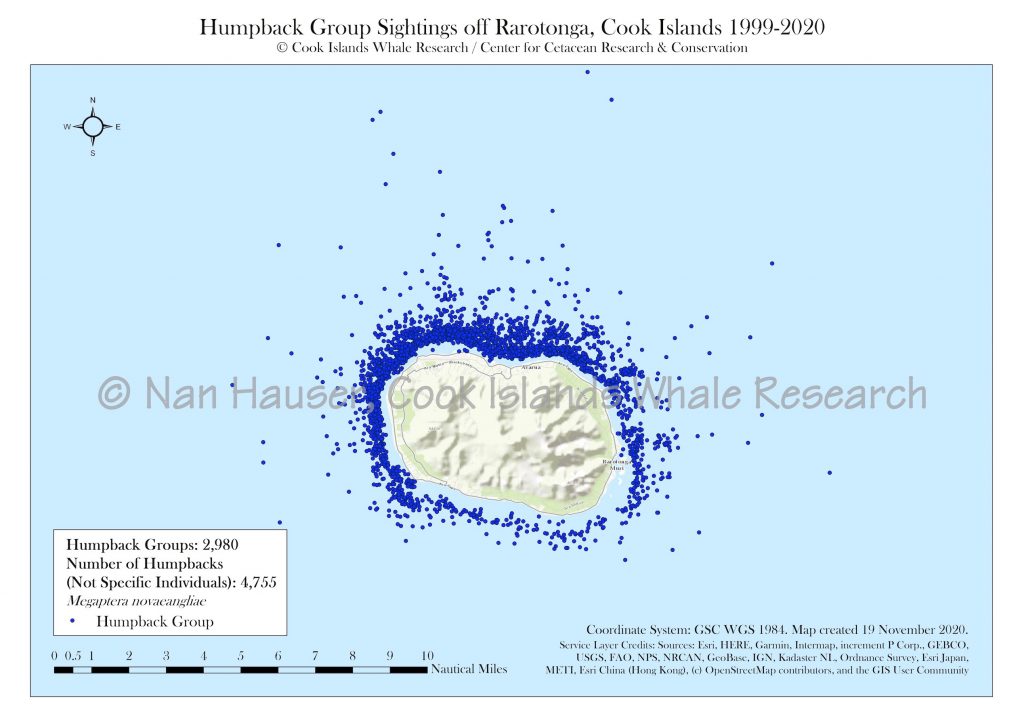
Between 1998 and 2020, CIWR has spent a total of 25,266 hours and 29 minutes surveying humpback whales over the course of 2,611 days. 9,750 hours and 26 minutes were spent at sea over the course of 1,587 of those days. CIWR has documented 4,862 humpback whales in 3,047 groups. We have had 1,611 singles; 775 pairs; 186 trios; 343 mother and calf pairs; and 170 mother, calf, and escort groups. 420 singers were also documented.
Photo-Identification
Identification photographs (photo-ID’s) are taken of the uniquely marked flukes, the left and right side of the dorsal fin, scarring and lateral pigmentation of each whale encountered.
Photo-ID’s are acquired using digital still cameras equipped with telephoto and zoom lenses. Distinctive markings and behaviour of whales are recorded with digital video cameras at the surface, and when necessary, underwater.
All images are digitized and data transcribed and subsequently integrated into databases. Photo-ID’s and video stills are entered into a Humpback Photo-ID Catalog to monitor individual whales and patterns of association.
Humpbacks passing through the corridor of the Cook Islands often dive without fluking, which is problematic for proper photo-ID shots of their flukes. In response to this low fluke rate, CIWR researchers photograph humpback tail flukes underwater when necessary, by holding a GoPro or Osmo Action camera over the side of the boat. Lateral pigmentation has been documented on a large percentage of the humpbacks observed in the Cook Islands.
Abundance of humpback whales in Oceania using photo-ID and microsatellite genotyping
Abundance and interchange of humpback whales in Oceania based on fluke photo-ID and DNA profiling
Acoustics
 CIWR researches the production of humpback song and cetacean vocalizations by using hydrophones and digital recorders to record and document the vocalizations of individual animals. CIWR gathers this acoustic data to better understand cetacean communication and culture.
CIWR researches the production of humpback song and cetacean vocalizations by using hydrophones and digital recorders to record and document the vocalizations of individual animals. CIWR gathers this acoustic data to better understand cetacean communication and culture.
Male humpback whales sing long and complex songs in the waters of the Cook Islands and other winter breeding grounds in the tropical oceans. Since male humpbacks usually sing during the breeding season, it is likely that the song relates to reproductive function. The song may serve to attract females, assert dominance among other males, maintain a distance between courting males, report information relating to the migration route and the feeding grounds, or many other possibilities that are unknown to us.
Song structure and form have been compared among the Cook Islands' humpbacks and songs recorded at other breeding grounds across the South Pacific and from around the world. Because humpback song varies geographically, comparative analysis of song and DNA samples from the singers are used to determine the stock identity of Cook Islands' humpbacks. Moreover, analysis of the phrases and singers' behaviour yields insights into the functional significance of the humpback song. Over the past 23 field seasons, we have recorded humpback whale songs every year and observed song exchange in the region. After University of Queensland acousticians analyzed SPWRC's 775 songs collected across Oceania, the data reveals song phrases passing from west to east, indicating horizontal cultural transmission of song. Over the past few years, we have had 2 to 3 distinct songs present over the whale season off Rarotonga, which poses a very interesting question of how song interchange is happening here in the Cook Islands.
Migratory convergence facilitates cultural transmission of humpback whale song
Dynamic horizontal cultural transmission of humpback whale song at the ocean basin scale
 Population and Behaviour
Population and Behaviour
CIWR documents the populations of humpback whales and other cetaceans that pass through the Cook Islands and observes the behaviour of individual animals.
Field data are documented using voice recorders, GPS, digital imaging and digital video. These include behaviours, individual whales present, geographic position, date, time, etc.
We observe and film both above and underwater behaviour with underwater cameras, GoPro’s, Osmo Action cameras, drones, 360° cameras and pole-cams.
Abundance of humpback whales in Oceania (South Pacific), 1999-2004
Occurrence & Habitat Use of Humpback Whales in the Cook Islands
Surface and Underwater Footage
Video footage is collected to study the behaviour of whales above and below the surface.
Underwater footage can also be used to obtain photo-identification images if these cannot be obtained at the surface, as is often the case with mother and calf pairs, as new mothers seldom show their flukes when they dive.
Underwater footage is obtained by either filming with an underwater camera from the side of the boat or while snorkeling.
Distribution
Using GPS data, including longitude and latitude coordinates, CIWR documents the distribution of all cetaceans that the team observes. These data are compared seasonally to understand habitat use in and among species and in relation to oceanographic variables and global climate change. We have documented every geographic coordinate for sightings from 1998 to 2020 and mapped these points through Geographic Information Systems (GIS).
 Satellite Tagging
Satellite Tagging
CIWR attaches satellite tags onto whales in order to test theoretical frameworks of animal navigation using empirical animal location data. This allows us to observe a predictive model for long-distance animal migration, as the whales migrate from Rarotonga to other areas in the Cook Islands and Oceania, and ultimately to Antarctica.
CIWR uses either a pole or pneumatic deployment device to apply the tags onto the whales, approximately at the base of the dorsal fin. These tags consist of a satellite transmitter attached to a stainless-steel anchoring system. The location of the tagged animal is transmitted to an Argos satellite and can be downloaded in nearly real time.
The knowledge of the humpback whale migration route from Oceania to Antarctica bolsters important efforts to minimize the spatial and temporal overlap between migrating whales and potential fishing gear. Previously  collected satellite tagging data will soon be used to enforce legislation prohibiting the deployment of long line fishing gear in the path of humpback whales migrating across Oceania and down to the Antarctic feeding grounds.
collected satellite tagging data will soon be used to enforce legislation prohibiting the deployment of long line fishing gear in the path of humpback whales migrating across Oceania and down to the Antarctic feeding grounds.
Straight as an arrow: humpback whales swim constant course tracks during long-distance migration
Route Fidelity during Marine Megafauna Migration
Movements of satellite monitored humpback whales from New Caledonia and the Cook Islands
Movements of satellite monitored humpbacks from the Cook Islands
Migratory destination of a humpback whale satellite tagged in the Cook Islands
Genetics
CIWR opportunistically and non-invasively collects animal bodily fluids and tissues from live or dead animals, including sloughed skin, biopsied skin, blubber samples, fecal samples, semen and bone.
Biopsies are frozen and shipped to collaborating universities for analysis. The DNA will be subsequently analyzed to determine the sex and relatedness of individuals. On a larger scale, samples are compared with those in existing databases to assess population identity of Cook Islands' humpbacks and genetic diversity in humpbacks worldwide.
Toxic Contaminants
The long lives and high lipid content in the blubber of humpback whales predispose them to be severely affected by exposure to chronic contaminant burdens. Since little research of this nature has been conducted, portions of the biopsy samples collected for genetic analysis will be frozen and later analyzed for toxic contaminant burden and biomarkers to evaluate the toxicological status of the Cook Islands' humpbacks.
Stable Isotopes
We use the identification of isotopic signature, the distribution of certain stable isotopes and chemical elements within chemical compounds gleaned from skin samples. This can be applied to a food web making it possible to draw direct inferences regarding diet, trophic level and subsistence. Variations in isotope ratios from isotopic fractionation are measured using mass spectrometry. This process separates the different isotopes of an element on the basis of their mass-to-charge ratio.
Using Mitochondrial DNA and Mixed Stock Analysis to Estimate Migratory Allocation
 Infrared Analysis and Multispectral Imaging
Infrared Analysis and Multispectral Imaging
In collaboration with University of Canterbury and Dr. Travis Horton, a study was conducted that observes humpback whales using the infrared spectrum. This was the first infrared analysis conducted on whales in a warm water environment. The hope of the project is to create an innovative method of identifying individual whales based on the signature of scarring patterns visible by infrared and a possible passive detection method for documenting the number of whales. We achieve this by using drones with infra-red cameras attached.
We use drones and multispectral imaging as a tool to measure the humpback whales’ biomedical conditions. Through infrared and multispectral imaging, we are able to take measurements of the whale’s body size, physiological conditions and vital signs (pulse, respiration and body temperature), which will help us monitor the health of these whales.
Through this project, we will achieve: 1) non-invasive monitoring of cetacean biomedical vital signs using drone-deployed thermographic sensors across a variety of behavioural states; 2) creating 3-D models of whale morphology and body condition at the individual level using drone-mounted stereoscopic multispectral (i.e. water-penetrating ‘coastal’ blue wavelength) cameras; 3) quantifying the relationship between the timing of long-distance return migration, whale condition, feeding behaviour and genetic markers.
Thermal Imaging and Biometrical Thermography of Humpback Whales
Humpback Response to Climate Change
Whales are incredible bio-indicators of climate change. They are affected more directly than many other species due to their migration patterns and feeding grounds at the poles.
One of the most obvious signs of climate change in the ocean is the change in arrival of species to certain areas. The humpback whales seem to be arriving to the Cook Islands later every year. With melting glaciers and changing water temperatures, the krill and herring that humpback whales feed on have had to move from their areas. The amount of krill in the Antarctic (the feeding ground of our humpback whales) has decreased by 70-80% since the 1970's.
We are striving to understand humpback response to environmental changes, such as changing primary productivities, water temperatures, conductivities and surface currents. We will investigate this mystery by 1) tracking humpback movements; 2) measuring humpback biomedical conditions using drones; 3) quantifying humpback multi-decadal age; 4) relating humpback behaviour and condition to environmental conditions and genetic markers.
Stable isotope studies will be used to understand humpback feeding strategies. We can do this with DNA samples, such as sloughed skin and fecal matter, to determine what the animals are eating and how their feeding strategies may be changing due to climate change. It will give us an indication of how they are or are not adapting to these changes.
Cook Islands Beaked Whale Survey
In 1998, CIWR began to survey and observe beaked whales in the waters of the Cook Islands and continues to collect data relating to the group structure. Sightings of beaked whales are extremely rare due to the fact that they favor deep water habitats, have long dive times, and exhibit unobtrusive surfacing behaviour. Accordingly, information is largely gathered from stranded or deceased animals.
Provisional data indicates that beaked whales rely on isolated critical habitat. The data collected from CIWR contributes to the Cook Island Marine Park Management Plan’s ability to identify and protect that critical habitat from potential threats such as long-line fishing, nets and intentional harm from foreign fishing boats.
In addition, because they communicate and navigate almost entirely using sound, beaked whales are sensitive to acoustic pollution. Threatening sources of acoustic pollution in marine environments may include oil prospecting, undersea mining, shipping noise and military sonar. As it has been communicated that foreign militaries have plans to use the Pacific for acoustic exercises in the future, CIWR is monitoring such activity due to the concern of the possible negative impacts of such activities on the Cook Islands' cetaceans.


 Resident Pod of Spinner Dolphins
Resident Pod of Spinner Dolphins
Spinner dolphin sightings have been documented around Rarotonga since 2000; however, it has been only since 2016 that CIWR has observed spinner dolphin pods to be resident to Rarotonga.
Spinner dolphins appear to have come and gone over the years. In the 23 years that CIWR has conducted research around Rarotonga, we have sighted dolphins in 12 of those years. We had no sightings in 1998-1999, 2002-2003, 2006-2007, 2009-2011 and 2014-2015.
In March 2016, we were alerted by the fishermen that there were dolphins spotted off the reef of the southwest side of the island. On observation, we found a pod of spinner dolphins mixing with Fraser's dolphins. After no dolphins sightings in 2014 or 2015, we were very pleased to see the spinners return. We have continued to observe spinner dolphins over the following years. In 2020, we had more spinner dolphin group encounters than ever before, with a total of 32 sightings!
We have photographed and filmed the dolphins as much as possible above water. We hope to enhance our cetacean catalogue by including the ID’s of these dolphins by photographing their dorsal fins at a right angle. In 1998 and 1999, photo ID’s were taken of the resident spinner dolphins off of Palmerston Atoll.
Cetacean Species Observed in the Cook Islands
- Humpback whale (Megaptera novaeangliae)
- Spinner dolphin (Stenella longirostris)
- Blainsville’s beaked whale (Mesoplodon densirostris)
- Antarctic minke whale (Balaenoptera bonaerensis) or
- Dwarf minke whale (Balaenoptera acutorostrata)
- Sperm whale (Physeter macrocephalus)
- Short-finned pilot whale (Globicephala macrorhynchus)
- Cuvier’s beaked whale (Ziphius cavirostris)
- Sei whale (Balaenoptera borealis)
- Fraser’s dolphin (Lagenodelphis hosei)
- Melon-headed whale (Peponocephala electra)
- Killer whale (Orcinus orca)
- False killer whale (Pseudorca crassidens)
- Pygmy sperm whale (Kogia breviceps)
- Fin whale (Balaenoptera physalus)
- Blue whale (Balaenoptera musculus)
- Bryde's whale (Balaenoptera edeni)
- Pygmy killer whale (Feresa attenuata)
- Omura's whale (Balaenoptera omurai)
- Rough-toothed dolphin (Steno bredanensis)
- Risso’s dolphin (Grampus griseus)
- Common dolphin (Delphinus delphis / frontalis)
- Bottlenose dolphin (Tursiops truncatus)
- Peale’s dolphin (Lagenorhynchus australis)
- Spotted dolphin (Stenella attenuata)
- Striped dolphin (Stenella attenuata)
- Dwarf sperm whale (Kogia simus)
For more information about our organisation’s benign research on whales, feel free to contact us.